Physicians
Learn more about our INSPIRE 2.0 clinical trial
Our Neuro-Spinal Scaffold is currently in clinical development for the treatment of acute thoracic spinal cord injury. The Neuro-Spinal Scaffold is focused on treating spinal cord injury directly at the epicenter of the injury.
At InVivo, we are deeply committed to working with physicians and health care professionals. If you are a physician interested in learning more about the Neuro-Spinal Scaffold, including clinical trial sites and a summary of the clinical trial protocol, please see below.
INSPIRE 2.0 clinical trial
The purpose of the INSPIRE 2.0 trial is to assess the overall safety and probable benefit of the Neuro-Spinal Scaffold for the treatment of neurologically complete thoracic traumatic acute spinal cord injury. INSPIRE 2.0 is designed to enroll ten patients each into one of two study groups: a treatment group and a control group. Patients in the control group will receive the standard of care, which is spinal stabilization.
INSPIRE 2.0 is a single blind study, meaning that the patients and assessors are blinded to treatment assignments.
The primary endpoint of the INSPIRE 2.0 trial is the proportion of patients achieving an improvement of at least one AIS grade at six months post-implantation. As per the protocol, the study will be deemed a success if the treatment group achieves 20% or greater improvement as compared to the control group. Assessments of AIS grade are at hospital discharge, three months, six months, 12 months and 24 months.
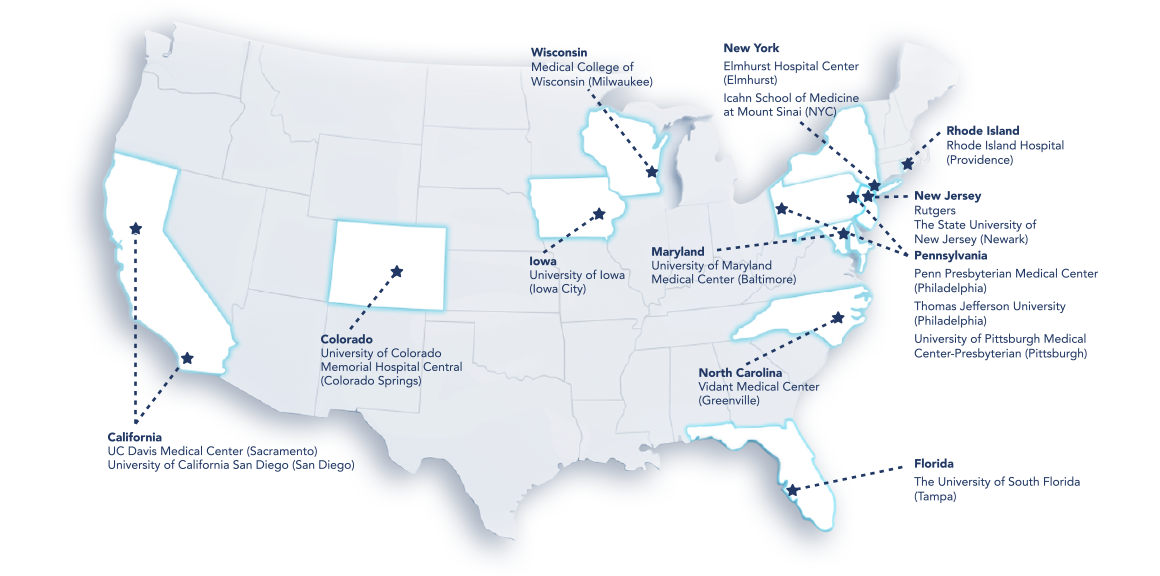
Trial locations
UC Davis Medical Center (Sacramento)
University of California San Diego (San Diego)
University of Colorado Memorial Hospital Central (Colorado Springs)
The University of South Florida, Tampa, FL
University of Iowa (Iowa City)
University of Maryland Medical Center (Baltimore)
Rutgers, The State University of New Jersey, Newark, NJ
Elmhurst Hospital Center (Elmhurst)
Icahn School of Medicine at Mount Sinai (NY)
Vidant Medical Center (Greenville)
Penn Presbyterian Medical Center (Philadelphia)
Thomas Jefferson University (Philadelphia)
University of Pittsburgh Medical Center-Presbyterian (Pittsburgh)
Rhode Island Hospital (Providence)
Medical College of Wisconsin (Milwaukee)
InVivo Therapeutics Holdings Corp. owns the copyright to the Training Video and Protocol Synposis above and reserves exclusive rights in each copyright. By accessing these materials, you hereby agree that any use of these materials shall strictly be for non-commercial, educational purposes only, and no permission or license is hereby granted for use of the materials, including the right to reproduce or distribute the materials.