Our Technology
Aiming to promote neural regeneration after spinal cord injury

Caution: Investigational device. Limited by federal law to investigational use.
Our investigational Neuro-Spinal Scaffold™ is implanted at the site of damage within one week of spinal cord injury with the goal of supporting neural regeneration and cell growth.
If left untreated, a cavity will typically develop at the injury site, cutting off transmission of neural signals from the brain to all body parts below the injury. The scaffold is designed to encourage functional cells to fill in that cavity. If achieved, this structural change within the spinal cord has the potential to improve sensory and motor function and lead to a better quality of life for patients.
Our investigational Neuro-Spinal ScaffoldTM
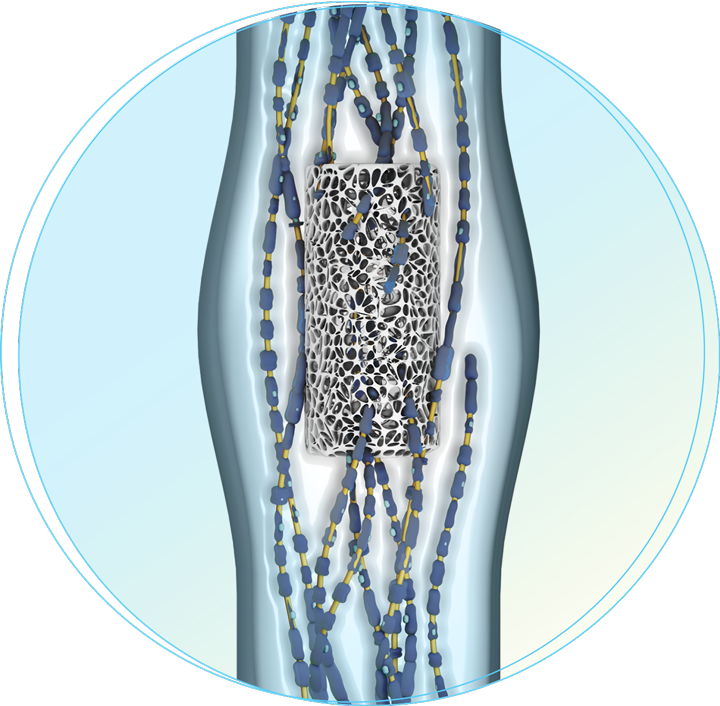
Our proprietary and investigational Neuro-Spinal Scaffold technology is composed of two biocompatible and bioresorbable polymers which together form a highly porous framework that is conducive to cellular attachment and growth.

The scaffold is surgically implanted into the gap in the spinal column that arises at the site of injury, aiming to create a neuropermissive matrix that allows cells which are critical to neuronal health to respond to the injury site and begin filling the cavity. The scaffold breaks down in the body over several weeks and is resorbed.
How the scaffold is designed to facilitate recovery
If achieved, a robust response can lead to improved outcomes for spinal cord injury patients by:


Preventing
development of an empty cavity at the injury site
Creating
an environment conducive to neural regeneration and tissue remodeling
Preserving
white matter and allowing for potential remyelination of axons
Visualizing improvement in an animal model
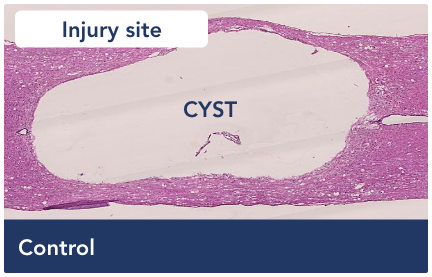
An empty cavity develops at the site of an untreated injury, cutting off communication from the brain.
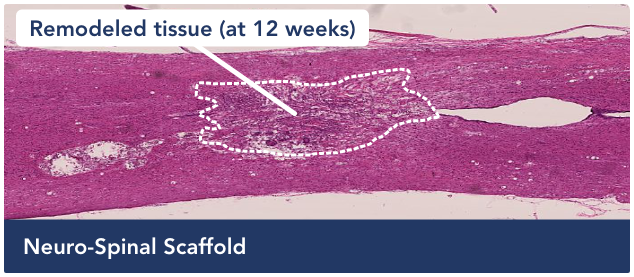
Treatment with the scaffold has encouraged tissue remodeling and neuronal growth.
The architecture of the spinal cord has not been restored to pre-injury status. However, the cyst is now filled with a neuropermissive matrix that allows cells, which are critical to neuronal health, to respond to the injury site and begin filling the cavity.
Existing Clinical Results
In its first human clinical study (INSPIRE 1.0), the Neuro-Spinal Scaffold was studied in 19 patients with acute thoracic spinal cord injury.
For more information on our clinical data, please see our corporate deck here.
Clinical trial underway
Based on the INSPIRE 1.0 data, InVivo has initiated the INSPIRE 2.0 clinical study. INSPIRE 2.0 is a 20-patient clinical study designed to assess the potential benefit of the Neuro-Spinal Scaffold in comparison to current standard of care by measuring improvement on the American Spinal Injury Association (ASIA) Impairment Scale (AIS Scale) at six months post-injury.
Visit clinicaltrials.gov for more information on our INSPIRE 2.0 Study.
Strong support in scientific literature
A robust collection of scientific publications, posters and presentations support the efficacy of the Neuro-Spinal Scaffold
Publications
Kim KD, Lee KS, Coric D, Harrop JS, Theodore N, Toselli RM | Acute implantation of a bioresorbable polymer scaffold in patients with complete thoracic spinal cord injury: 24-month follow-up from the INSPIRE study | Neurosurgery | 2022
Papadopoulos MC, Saadoun S | Letter to the Editor. The INSPIRE studies for spinal cord injury | J Neurosurg Spine | 2021
Kim KD, Lee KS, Coric D, Chang JJ, Harrop JS, Theodore N, Toselli RM | A study of probable benefit of a bioresorbable polymer scaffold for safety and neurological recovery in patients with complete thoracic spinal cord injury: 6-month results from the INSPIRE study | J Neurosurg Spine | 2021
Aimetti AA, Kirshblum S, Curt A, Mobley J, Grossman RG, Guest JD | Natural history of neurological improvement following complete (AIS A) thoracic spinal cord injury across three registries to guide acute clinical trial design and interpretation | Spinal Cord | 2019
Guest JD, Moore SW, Aimetti A, Kutikov AB, Santamaria AJ, Hofstetter CP, Ropper AE, Theodore N, Ulich TR, Layer RT | Internal decompression of the acutely contused spinal cord: differential effects of irrigation only versus biodegradable scaffold implantation | Biomaterials | 2018
Dukes EM, Kirshblum S, Aimetti AA, Qin SS, Bornheimer RK, Oster G | Relationship of American Spinal Injury Association Impairment Scale grade to post-injury hospitalization and costs in thoracic spinal cord injury | Neurosurgery | 2018
Layer RT, Ulich TR, Coric D, Arnold PM, Guest JD, Heary RH, Hsieh PC, Jenkins AL, Kim KD, Lee KS, Masuoka LK, Neff KM, Ray WZ, Theodore N, Fehlings MG | New clinical-pathological classification of intraspinal injury following traumatic acute complete thoracic spinal cord injury: postdurotomy/myelotomy observations from the INSPIRE trial | Neurosurgery | 2017
Slotkin JR, Pritchard CD, Luque B, Ye J, Layer RT, Lawrence MS, O'Shea TM, Roy RR, Zhong H, Vollenweider I, Edgerton VR, Courtine G, Woodard EJ, Langer R | Biodegradable scaffolds promote tissue remodeling and functional improvement in non-human primates with acute spinal cord injury | Biomaterials | 2017
Theodore N, Hlubek R, Danielson J, Neff K, Vaickus L, Ulich TR, Ropper AE | First human implantation of a bioresorbable polymer scaffold for acute traumatic spinal cord injury: A clinical pilot study for safety and feasibility | Neurosurgery | 2016
Pritchard CD, Slotkin JR, Yu D, Dai H, Lawrence MS, Bronson RT, Reynolds FM, Teng YD, Woodard EJ, Langer RS | Establishing a model spinal cord injury in the African green monkey for the preclinical evaluation of biodegradable polymer scaffolds seeded with human neural stem cells | J Neurosci Methods | 2010
Teng YD, Lavik EB, Qu X, Park KI, Ourednik J, Zurakowski D, Langer R, Snyder EY | Functional recovery following traumatic spinal cord injury mediated by a unique polymer scaffold seeded with neural stem cells. | Proc Natl Acad Sci U S A | 2002
Abstracts
Kim KD, Lee KS, Harrop JS, Coric D, Theodore N, Toselli RM | Timing of neurological recovery in patients with acute thoracic AIS A SCI implanted with an investigational Neuro-Spinal Scaffold™: results of the INSPIRE study | CNS Annual Meeting; September 12–16, Miami, FL (conference cancelled; abstract published) | 2020
Kim KD, Lee KS, Chang JJ, Toselli RM | Neuro-Spinal Scaffold™ implantation in patients with acute traumatic thoracic complete spinal cord injury: twenty-four month results from the INSPIRE study | CNS Annual Meeting; October 19–23, San Francisco, CA | 2019
Kim KD, Lee KS, Coric D, Theodore N, Toselli RM | Twelve month results from the INSPIRE study of the investigational Neuro-Spinal Scaffold™ in acute thoracic complete spinal cord injury | AANS Annual Meeting; April 13–17, San Diego, CA | 2019
Lee KS, Coric D, Theodore N, Kim KD | Complete 6-month primary endpoint results from the INSPIRE study of the investigational Neuro-Spinal Scaffold™ in acute thoracic complete spinal cord injury | AANS Annual Meeting; October 6–10, San Diego, CA | 2018
Lee KS, Boyer PJ, Hsieh PC, Hurth KM, Guest JD, Hofstetter CP, Aimetti A, Layer RT, Moore SW, Ulich TR | Histopathology of necrotic spinal cord tissue exudate collected during surgical implantation of a biodegradable scaffold following acute spinal cord injury: pre-clinical and clinical findings | CNS Annual Meeting; October 7–11, Boston, MA | 2017
Moore SW | The Neuro-Spinal Scaffold promotes tissue remodeling, axonal sprouting, and Schwann cell myelination following acute spinal cord contusion injury in rats | Society for Neuroscience (SFN) Annual Meeting; November 12–16, San Diego, CA | 2016
Aimetti AA | Translation of biomaterial-based therapies for the treatment of spinal cord injury: the Neuro-Spinal Scaffold™ and bioengineered neural trails | Working 2 Walk Science & Advocacy Symposium; October 28–29, Minneapolis, MN | 2016
Aimetti AA | Translation of biomaterial-based therapies for the treatment of spinal cord injury: the Neuro-Spinal Scaffold™ and bioengineered neural trails™ | Symposium on Biomaterials Science; October 24–25, Iselin, NJ | 2016
InVivo Therapeutics developing innovative products for spinal cord injury | Cell & Gene Meeting on the Mesa; October 5–7, La Jolla, CA | 2016
Theodore N | High ASIA Impairment Scale conversion rate following scaffold Implantation in acute thoracic complete AIS A spinal cord injury (SCI): potential mechanisms | CNS Annual Meeting; September 24–28, San Diego, CA | 2016
Fehlings MG | Clinical-pathological assessment of severe (AIS A) traumatic acute thoracic spinal cord injury: post-durotomy/myelotomy observations from the INSPIRE trial | CNS Annual Meeting; September 24–28, San Diego, CA | 2016
Heary RF, Barry M, Coric D, Kim KD, Hsieh PC, Ray WZ, Theodore N | MR Neuroimaging does not accurately identify intraparenchymal cavities in the spinal cord following AIS A thoracic spinal cord injuries | CNS Annual Meeting; September 24–28, San Diego, CA | 2016
Neff KM | Clinical trials update for 2016 – the INSPIRE study: Neuro-Spinal Scaffold™ | International Spinal Cord Society (ISCoS) Annual Meeting; September 14–16, Vienna, Austria | 2016
Layer RT, Moore SW, Kutikov AB, Podell P, Aimetti AA, Ulich TR | Biodegradable Neuro-Spinal Scaffold promotes neuropermissive remodeled tissue following spinal contusion injury in rats | National Neurotrauma Society (NNS) Annual Symposium; June 26–29, Lexington, KY | 2016
Theodore N, Coric D, Hsieh P, Kim KD | Neurologic outcome following implantation of a Neuro-Spinal Scaffold™ into the lesion cavity in acute thoracic complete spinal cord injury: results of a pilot study | Spine Summit; March 16–19, Orlando, FL | 2016
Masuoka L, Neff K, Ulich T, Aimetti AA | Novel surgical procedure to implant an investigational Neuro-Spinal Scaffold following myelotomy for treatment of acute traumatic spinal cord injury: a safety and feasibility study | CNS Annual Meeting; September 26–30, New Orleans, LA | 2015
Aimetti AA, Layer RT, Moore SW, Ulich TR | Biodegradable Neuro-Spinal Scaffold preserves macroscopic spinal cord architecture and allows for neural regeneration at the epicenter of spinal cord injury | Tissue Engineering & Regenerative Medicine International Society (TERMIS) World Congress; September 9, Boston, MA | 2015
Layer R, Aimetti A, Podell P, Moore S, Ulich T | Biodegradable Neuro-Spinal Scaffold preserves spinal cord architecture following spinal contusion injury in rats | National Neurotrauma Society (NSS) Symposium; June 28–July 1, Santa Fe, NM | 2015
Ropper AE, Hlubek R, Danielson J, Theodore N | First human implantation of a polymer scaffold for traumatic spinal cord injury: a clinical pilot study for safety and feasibility | AANS Annual Meeting; May 2–6, Washington, DC | 2015
Guest JD, Hofstetter C, Aimetti AA, Layer R, Hou J, Moore S, Ulich T | Reduction of intraparenchymal pressure after porcine acute spinal cord contusion injury by Neuro-Spinal ScaffoldTM implantation | AANS Annual Meeting; May 2–6, Washington, DC | 2015
Layer RT | Development and clinical translation of biomaterial technologies for spinal cord injury | 4th International Neural Regeneration Symposium (INRS); September 6–8, Nanjing, China | 2014